|
Combining the in-phase orbitals results in a bonding orbital. One contains the axis, and one contains the perpendicular. Combining the out-of-phase orbitals results in an antibonding molecular orbital with two nodes. All the elements in the second period before oxygen have the difference in energy between the 2s and 2p orbital small enough, so that s-p mixing (combination) can occur lowering the energy of the (2s) and (2s) and increasing the energy of the (2p) and (2p) molecular orbitals. Side-by-side overlap of each two p orbitals results in the formation of two π molecular orbitals. This phenomenon is explained by s-p mixing. For the out-of-phase combination, there are two nodal planes created, one along the internuclear axis and a perpendicular one between the nuclei.įigure 7.7.6. Electrons in this orbital interact with both nuclei and help hold the two atoms together, making it a bonding orbital. In molecular orbital theory, we describe the \pi orbital by this same shape, and a \pi bond exists when this orbital contains electrons. In valence bond theory, we describe π bonds as containing a nodal plane containing the internuclear axis and perpendicular to the lobes of the p–\pi orbitals, with electron density on either side of the node. The side-by-side overlap of two p orbitals gives rise to a pi (\pi) bonding molecular orbital and a \pi* antibonding molecular orbital, as shown in Figure 7.7.6. Combining wave functions of two p atomic orbitals along the internuclear axis creates two molecular orbitals, σp and σ∗p. Just as with s-orbital overlap, the asterisk indicates the orbital with a node between the nuclei, which is a higher-energy, antibonding orbital.įigure 7.7.5. There is an \ce^* (antibonding) (read as “sigma-p-x” and “sigma-p-x star,” respectively). Furthermore, as there are two unpaired electrons in an Oxygen molecule hence it is paramagnetic.This electronic structure adheres to all the rules governing Lewis theory. double bonds formed between two oxygen atoms (O=O). As electrons are also present in antibonding molecular orbitals so weak bonds will be formed.īond order = Number of electrons in BMO – Number ofĪs the bond order in Oxygen is 2 so two bonds i.e. Out of eight electrons, six go to bonding molecular orbitals and two to the antibonding molecular orbitals. The electron filling in these molecular orbitals follows Aufbau, Pauli exclusion principle and Hund’s rule. π2py and π2pz, while two antibonding molecular orbitals i.e. The other four p-atomic orbitals (two from each oxygen) atom combines to give four molecular orbitals, two bonding molecular orbitals i.e. Two p-atomic orbitals (one from each oxygen) atom combine to form two molecular orbitals, the bonding molecular orbital σ2px and antibonding molecular orbital σ*2px. electronic configuration of oxygen mo diagram of oxygen Oxygen atom has electronic configuration 1s2, 2s2, 2p4. Now let’s understand !! MO Diagram of O2 molecule mo diagram of oxygen electronic configuration of oxygen 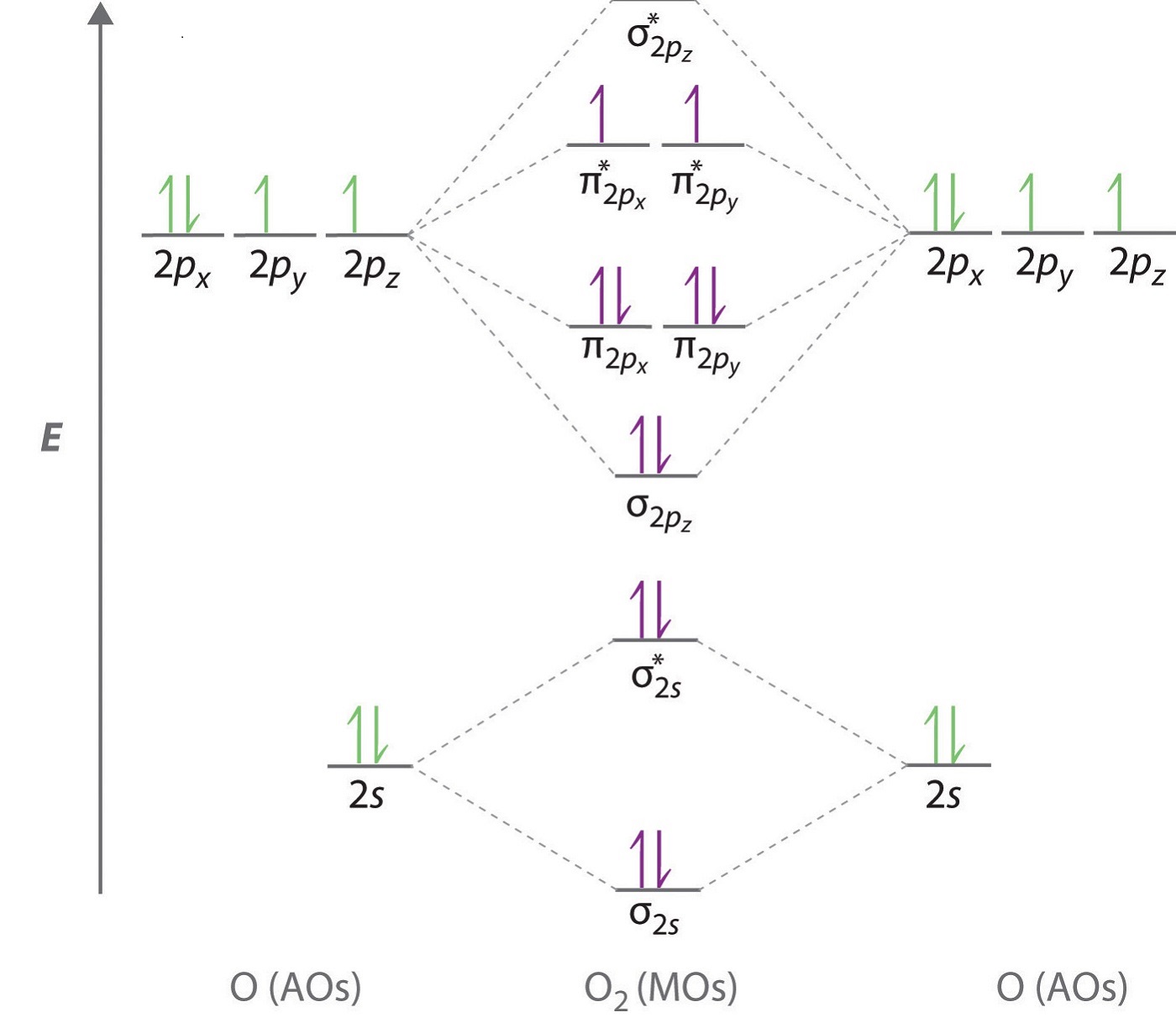
The total energy of the MOs is always equal to the total energy of combining atomic orbitals. Half of the molecular orbitals (MOs) having energy lower than the atomic orbitals are called bonding molecular orbitals (BMOs) while half of the molecular orbitals (MOs) having energy higher than the atomic orbitals are called Anti-bonding molecular orbitals (ABMOs) 3. Molecular orbital (MO) theory explains the construction of a molecular orbital diagram on the basis of the following main points.Ītomic orbitals(AOs) linearly combine with each other to form an equal number of molecular orbitals (MOs).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
